
Article Contents
Article ID: CM2601107005
Digital Health Interventions and Virtual Care Models to Optimise Resource Allocation and Patient Outcomes in Post-Pandemic Healthcare Systems
⬇ Downloads: 13
Received: 11 January, 2026
Accepted: 15 March, 2026
Revised: 29 February, 2026
Published: 31 March, 2026
Abstract:
The COVID-19 pandemic accelerated the adoption of digital health interventions (DHIs) and virtual care models to mitigate workforce shortages, bed scarcity, and rising demand. While early evidence suggested potential to reduce hospital strain and improve outcomes, the effectiveness, equity, and sustainability of these approaches in the post-pandemic recovery remain unclear. A systematic review with narrative synthesis and contextual evidence mapping methodology in line with the PRISMA 2020 guidelines was applied to search three databases, PubMed, Scopus, and Web of Science, with 2020-2025 as the search period. Included studies were peer-reviewed principal studies of digital health interventions (DHIs) and individual reviews of telemedicine, mobile health (mHealth), remote patient monitoring, and virtual wards that reported on resource allocation or patient outcomes. Evidence demonstrated that structured, specialist-led models (e.g., heart failure virtual wards) reduced hospitalisations and mortality, while remote monitoring programmes showed mixed results due to low enrolment and inconsistent implementation. Telehealth enhanced continuity but widened equity gaps in disadvantaged populations. Virtual wards and hospital-at-home models reduced length of stay and improved patient well-being in well-resourced systems, but often redistributed demand rather than reducing it. AI-based models accurately predicted hospitalisation and mortality but raised concerns over bias, generalisability, and transparency. DHIs offer significant potential for resilience and efficiency but require equitable access, implementation fidelity, interoperability, and ethical safeguards. Policymakers, providers, and researchers must collaborate to ensure sustainable integration into routine healthcare.
Keywords: Digital health intervention, virtual care models, resource allocation, patient outcomes, and post-pandemic healthcare.
1. INTRODUCTION
The coronavirus pandemic has led to massive changes in healthcare provision, putting substantial pressure on healthcare systems worldwide. In balancing acute care and chronic disease, healthcare organisations have found themselves short of workforce, bed capacity, and equipment, undermining their ability to maintain patient outcomes and safety. Therefore, the crisis accelerated the adoption of digital health tools, such as telemedicine, remote patient monitoring, and AI-based decision support systems, which required a rapid response to new demands [1]. Digital health interventions (DHIs), as a category that includes telecopying services, mobile health (mHealth) applications, virtual wards, and remote monitoring devices, have become the cornerstone of continuity and efficiency in care provision during the pandemic. Such technologies have relieved conventional healthcare systems by enabling remote diagnosis, surveillance, and treatment [2]. Therefore, DHIs will be able to optimise resource allocation and ensure the well-being of patients and providers. Specifically, virtual care models, including the virtual hospital and hospital-at-home models, provide hospital-level care at patients’ homes, thus reducing length of stay in inpatient settings and correcting resource shortages [3].
The post-pandemic period necessitated evaluating the contribution and use of digital innovations to support long-term service delivery. There were workforce shortages, a lack of beds, and delays in delivering treatments, which had already been pressing on traditional systems but were eliminated thanks to DHIs and virtual care models. Without such interventions, the healthcare system would probably have seen patient backlogs escalate and mortality rates increase, possibly linked to inaction and further burdening hospitals. Implementations of remote monitoring systems and telemedicine aim to ensure continuity of care and prevent systemic breakdowns in institutions where telemedicine is applied. However, when they are introduced into routine practice, they must be meticulously grounded in efficacy, equity, and sustainability. These technologies can produce quantifiable changes in patient outcomes, strengthen healthcare systems’ resilience, and address systemic gaps far beyond the ability to respond to crises. The prudent use of telemedicine could lead to high patient attendance rates and positive health outcomes across a range of chronic illnesses. The initial evidence suggests that telehealth interventions are effective and cost-optimising in the clinic [5]. Besides digital health interventions have had a positive impact on the population health metrics and clinician satisfaction. However, the viability of implementation, data integration, and evidence transparency has not been fully discussed [6]. The impact of digital health interventions (DHIs) and virtual care models on resource use and patient outcomes remains poorly understood. Indeed, there are examples of such analysts arguing that MH and monitoring technologies can reduce hospital visits and length of stay [6][7]. Nevertheless, these studies are often limited by small sample sizes, short-term follow-up, and incomplete enrolment, thus limiting the applicability of the findings. These restrictions highlight the importance of a rigorous review to analyse the evidence base critically. However, extensive evaluations of effectiveness, especially in a post-pandemic recovery strategy, require additional standardisation [1]. Similarly, models like the PICOTS ComTeC offer insight into the design of digital health research, but further research is needed to implement the resource and outcome metrics into system-level performance [9].
This systematic literature review (SLR) therefore aims to critically evaluate previous studies (quantitative and qualitative) on the evolution of digital health before and after the pandemic, published between 2020 and 2025. The review discusses the impact of digital health interventions and virtual care models on resource allocation: length of stay, bed utilisation, staffing needs, and cost-effective usage in the post-COVID-19 period in the healthcare system. It explores evidence regarding patient outcomes, including clinical effectiveness, patient satisfaction, readmission rates, and mortality. Moreover, it identifies structural and implementation factors that influenced the success of digital health interventions in strengthening healthcare system resilience to future shocks. The review aims to clarify both the measurable benefits of digital and virtual strategies in optimising resources and patient outcomes, and to identify the facilitators and inhibitors that mediate their adoption. The insights have the potential to guide policymakers, healthcare leaders, and academics on how to develop sustainable, digitally empowered care models that improve system resilience in the post-pandemic era. This review is framed as a healthcare system–level narrative synthesis of digital care models for post-pandemic system resilience.
2. Methods
2.1. Research Design
This study employed a systematic literature review (SLR) design to critically synthesise evidence on the role of digital health interventions (DHIs) and virtual care models in optimising healthcare resource allocation and patient outcomes in the post-pandemic era. A systematic review was selected over a narrative approach because it offers a rigorous, transparent, and replicable methodology that minimises selection bias and enhances the reliability of findings [10]. The review followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA 2020) framework, thereby enabling systematic identification, screening, and synthesis of eligible research [11]. This research is further complemented by a narrative synthesis of the included primary studies and a contextual evidence mapping of selected secondary and policy-oriented sources.
2.2. Literature Search Strategy
The extensive search was carried out in three interdisciplinary databases, PubMed, Scopus, and Web of Science, which included the articles and journals of 2020-2025. Boolean operators and keywords were adapted to each database. A mutual search string used in all databases included (“digital health” OR telemedicine OR telehealth OR “remote patient monitoring” OR “virtual ward*” OR “hospital at home”) AND (“resource allocation” OR “healthcare utilization” OR “hospital capacity”) AND (“patient outcome*” OR mortality OR readmission*) AND (“COVID-19” OR “post-COVID” OR “post-pandemic”). For detailed searches, see Appendix B below. Searches were restricted to peer-reviewed English-language studies involving human participants. Primary empirical studies formed the main synthesis set, while selected reviews, protocols, and policy sources were retained separately for contextual evidence mapping.
2.3. Inclusion and Exclusion Criteria
Primary empirical studies included for synthesis; reviews/protocols/policy excluded from synthesis, except for a few. However, only primary studies contributed to effect estimates. The studies between 2020 and 2025 that investigated digital health or virtual care interventions, such as telemedicine, mHealth, remote patient monitoring, and virtual wards. Eligible studies involved human participants and reported outcomes related to resource allocation (length of stay, utilisation, costs, efficiency) and/or patient outcomes (mortality, readmissions, satisfaction, health indicators). Primary empirical studies constituted the main evidence base for synthesis, while selected reviews, protocols, and policy sources were retained separately as contextual evidence and were not used for effect estimation. Excluded were non-English publications, protocols, opinion pieces, policy briefs, conference abstracts, and studies limited to animal or laboratory models.
2.4. Data Screening
The screening followed a multi-stage PRISMA-guided process. Screening was conducted in two stages: first at the title and abstract level, then at the full-text eligibility level. A total of 214 records were identified from PubMed (n = 91), Web of Science (n = 72), and Scopus (n = 51). After eliminating duplicate entries and automatically eliminating records that fell outside the set temporal limits (n = 76), 138 records were left to undergo title- and abstract-level screening. Of these, 83 were filtered out because irrelevant (48 titles and abstracts, 13 full-text screening, 15 not within the scope of this review, and 7 non-peer-reviewed sources). Of the 55 remaining reports, the full text was also to be searched; if 12 were not downloaded due to a lack of full texts, at most, only conference abstracts were available. Therefore, 43 full texts were used to evaluate eligibility, and 18 articles were filtered at this point (5 were not about COVID-19, and 13 were not about DHIs). A total of 25 studies that met all inclusion criteria were included in the final synthesis; otherwise, studies not directly related to health-system restructuring due to COVID were excluded. Title/abstract screening and full-text eligibility assessment were conducted independently by two reviewers, with disagreements resolved through discussion.
2.5. Data Extraction
A structured Excel template was used to extract the data, including the author, year, country, study design, sample size, type of DHI, and outcome measures. Resource allocation was conceptualised as capacity, workforce, and cost deployment decisions in healthcare systems, with capacity outcomes including bed-days, length of stay, and hospital utilisation; workforce outcomes including clinician time and staffing workload; and financial outcomes including cost per episode and cost per bed-day. The predictive models were considered planning tools to assist decision-making, not mechanisms for direct savings. Two independent reviewers extracted data, and disagreements were resolved by consensus. Relative risks, odds ratios, mean differences, and 95 per cent confidence intervals were provided where appropriate; the direction of the effect was recorded narratively.
2.6. Conceptual organising framework
To plan synthesis at the health-systems level, the study’s findings were analysed using the input-process-output-outcome framework. The inputs included staffing, digital infrastructure, devices, and implementation resources; the processes included patient selection, monitoring intensity, escalation pathways, and integration into the clinical workflow; the outcomes included mortality, patient experience, readmissions, equity, and system resilience. There is an intersection between each type of intervention and this framework, allowing the organisation of an otherwise heterogeneous body of study through the systematic comparison of the narratives. Primary empirical studies informed the analytical synthesis, and contextual evidence sources were used to support understanding of policy and implementation contexts.
2.7. Data Analysis
The analysis method was a manual thematic synthesis based on the approach presented in the journal article [12]. Synthesis clusters are focused on five domains close to one another, including but not limited to the effects on resource allocation (e.g. length of stay, hospital capacity, cost-efficiency), effects on patient outcomes (e.g. mortality, hospitalisation readmissions, patient satisfaction, clinical improvements), and contextual enabling or hindering factors (e.g. digital infrastructure, equity issues, staff adoption). Codes were iteratively grouped into descriptive themes corresponding to the input–process–output–outcome framework. Themes were compared across studies and interpreted in relation to study design and methodological quality. Thematic clustering was able to integrate measure quantities and qualitative information.
2.8. Quality Assessment
The methodological quality of all included studies was evaluated using the Critical Appraisal Skills Programme (CASP), MMAT, and JBI checklists. It provides structured guidance for assessing validity, relevance, and rigour across qualitative and quantitative designs [7]. Overall, the quality of the evidence was high or moderate and varied significantly due to study design, methodological transparency, and control of confounders. High-quality evaluations were most common in large systematic reviews of digital interventions and economic frameworks (e.g., Gentili et al. [27]; Benedetto et al. [33]; Zakhiyah et al. [28]). Further, well-powered comparative and device-based RCT analyses showed strong internal validity [24]. Virtual ward and remote monitoring cohorts were featured with moderate-quality evidence from retrospective designs, selection bias, and inadequate adjustment for illness severity, which made it difficult to draw causal conclusions [8,15,26]. Although the external validity was well-founded in the context of real-world implementation studies, a number of them lacked standardised outcome measures. It relied on descriptive data, limiting opportunities for generalisation, as noted by Vo et al. [30] and Jalilian et al. [32]. The lack of risk-of-bias analysis restricted the use of policy commentary and scoping analysis by Oliver [25], Islam et al. [29], and Santos et al. [34] as suitable choices to their purposes (See Appendix A). Systematic reviews and economic syntheses were appraised using the CASP Systematic Review Multiple appraisal tools (CASP, MMAT, and JBI) were used to ensure design-specific, methodologically appropriate quality assessment across heterogeneous study types. Checklist for evaluating search completeness, risk of bias assessment, data synthesis, and applicability to healthcare settings. High-quality reviews (e.g., Shi et al., Gentili et al., Benedetto et al.) were considered robust when they demonstrated transparent methods, comprehensive searches, and appropriate synthesis. In contrast, protocols and scoping reviews were rated lower, as they lacked outcome synthesis or formal risk-of-bias assessment. They were therefore used only for contextual or mapping purposes rather than evidential weighting (See Appendix A). Analogues were used to assess cohort and diagnostic studies using the Critical Appraisal Skills Program (CASP) tools, randomised controlled trials using the Joanna Briggs Institute (JBI) methodology, and the synthesis of evidence using the CASP systematic review checklist. Appraisal criteria were tailored to the study design, and systematic reviews were evaluated for search completeness, bias appraisal, and synthesis rigour, rather than for experimental control.
2.9. Ethical Consideration
The study relied exclusively on secondary analysis of published literature and did not involve human participants or identifiable patient data. As such, institutional ethical approval was not required. However, all efforts were made to ensure ethical integrity, including accurate citation, avoidance of plagiarism, and transparent reporting in accordance with PRISMA guidelines.
3. Analysis and Discussion
Effect sizes across heterogeneous study designs and outcome definitions are not directly comparable; therefore, findings are synthesised narratively by domain.
3.1. Virtual Wards and Hospital-at-Home Models
The rapid expansion of virtual wards (VWs) and hospital-at-home (HAH) models during the COVID-19 pandemic revealed mixed findings. Van Goor et al. [14] conducted an RCT that showed that remote hospital care reduced hospital stays by 1.6 days (see Table 1A). However, there was no significant increase in hospital-free days within 30 days, indicating that overall care responsibility increased. Remote hospital care reduced index length of stay by a mean difference (MD) of −1.6 days (95% CI −2.4 to −0.8; p<0.001), but did not increase hospital-free days at 30 days (MD = 1.7 days; 95% CI −0.5 to 4.2; p=0.112), and was associated with a longer total duration of hospital responsibility (MD = +4.1 days; 95% CI 0.5 to 7.7; p=0.028), demonstrating a shift rather than a reduction in care burden. Zychlinski et al. [15] reported more optimistic results, with telemedicine-controlled HAH reducing both length of stay (LOS) and mortality rates for COVID and non-COVID patients, along with no increase in readmissions. Median length of stay was 2 days shorter in the home model for both COVID-19 and non-COVID patients (median difference; 95% CI 1–3 days; p=0.008 and p<0.001, respectively), while 30-day readmission was not significantly different (COVID-19: OR = 1.00, 95% CI 0.49–2.04; non-COVID: OR = 0.70, 95% CI 0.39–1.28). In contrast, 30-day mortality was lower in hospital-at-home patients (COVID-19: OR = 0.34, 95% CI 0.11–0.86; non-COVID: OR = 0.38, 95% CI 0.14–0.90) Ward et al. [8] demonstrated that home oxygen therapy, within a virtual ward, resulted in low 30-day readmission (3%) and mortality (1%) rates. However, this study was limited by its single-centre design and patient selection bias. In contrast, Beaney et al. [13] found that the Oximetry @home program in England showed no mortality benefit and increased hospital and critical care admissions, with only 2.5% of eligible patients enrolled, severely diluting any potential benefits. These findings highlight significant variation in the effectiveness of virtual care, with infrastructure and patient engagement being key determinants of success.
Recent review evidence and implementation studies indicate that the success of virtual wards (VWs) and hospital-at-home (HAH) still heavily relies on context, scalability, and the standardisation of care. Although large syntheses, including Shi et al. [19], documented decreased length of stay and similar or even lower readmission rates than in hospital care, in real-world assessments, these benefits are not always associated with a decrease in overall healthcare demand. As an illustration, van Goor et al. [14] showed a decrease in index length of stay but did not see a decrease in hospital-free days, indicating that it is not that the responsibility has been shifted, but that the load has been shifted. Implementation assessments, such as Vo et al. [30], also demonstrated that only a sufficient staffing of the services and a digital supporting environment will lead to the implementation of bed-day savings, whereas a critical look at the policy by Oliver [25] cautioned them about the risk of creating a promising naivety and creating clinical risk when the implementation of virtual wards is poorly resourced. Economic reviews like that by Jalilian et al. [32] support this warning and show that even though VWs may liberate capacity, the price per bed-day might be higher than the conventional inpatient treatment under unfavourable staffing or occupancy.
3.2. Remote Monitoring and Resource Optimisation
The remote monitoring turned out to be an essential approach during the years of the COVID-19 pandemic, aimed at reducing the number of patients in hospitals and improving resource management. The evaluation of the COVID Oximetry @home program was performed by Beaney et al. [13] and was based on a national stepped-wedge design, which did not show any significant decrease in mortality, but did decrease the number of visits to the hospital. At population level, implementation of the COVID Oximetry@home programme was not associated with a reduction in mortality and showed higher service utilisation, with increased odds of emergency department attendance (OR = 1.12; 95% CI 1.06–1.18), emergency admission (OR = 1.12; 95% CI 1.05–1.20), and critical care admission among those admitted (OR = 1.24; 95% CI 1.05–1.47), reflecting increased downstream demand rather than resource substitution. Ward et al. [8], in their turn, focused on a single-centre virtual ward where COVID-19 patients are in need of oxygen support, and their readmission and mortality rates were low. In a real-world virtual ward providing home oxygen, 30-day outcomes showed 3.4% readmission (5/147) and 1.4% mortality (2/147), with no statistically significant difference between patients discharged with or without oxygen, indicating safety in carefully selected patients but without comparative effect estimates against standard inpatient care. Similarly, van Goor et al. [14] reported that telemedicine-supported remote hospital care for COVID-19 patients resulted in reduced hospital stays but no increase in “hospital-free days”. Zychlinski et al. [15] further demonstrated that hospital-at-home models lowered both length of stay and mortality, highlighting the benefits of remote care in well-resourced settings. Despite these positive outcomes, the evidence indicates that the success of remote monitoring is contingent upon patient selection, program scale, and consistent implementation.
Table 1. summarises the included characteristics and key outcomes of the selected studies.
Table 1(A). Included primary studies.
Author/Year | Aim of the Study | Methodology | Treatment / Intervention | Findings of the Study | Quality Appraisal Outcome |
Beaney et al., 2022 | To assess the population-level impact of COVID-19 Oximetry @home on mortality and healthcare utilisation in England. | National stepped-wedge design analysis of routine data. | Pulse Oximetry remote monitoring for COVID-19 patients. | It showed no mortality benefit and increased emergency department attendance, hospital admissions, and critical care use, but had a limited effect on mortality; low enrolment diluted the outcomes. | Moderate quality; robust national design, but low enrolment and variation in implementation limit the validity of impact. |
Sankaranarayanan et al., 2024 | To evaluate outcomes of telehealth-aided outpatient management of acute heart failure in a virtual ward. | Propensity-score matched cohort study (HF virtual ward vs. standard care). | Telehealth-guided outpatient management with IV furosemide. | Virtual ward significantly reduced re-hospitalisations and mortality at 1, 3, 6, and 12 months compared to standard care. | High quality; strong propensity-matching and outcome measures, but the specialist setting limits generalisability. |
Tariq et al., 2021 | To develop AI-based models predicting COVID-19 hospitalisation and resource use. | Retrospective cohort (3,194 patients across 12 centres, EMR data). | Fusion AI model integrating demographics, comorbidities, medications, and lab results. | The early fusion model predicted hospitalisation risk with an 84% F1-score; cardiovascular history and demographics were strong predictors. | Moderate–high quality; strong AI modelling with a large dataset, yet limited external validity due to regional data bias. |
Ward et al., 2022 | To evaluate the virtual ward oxygen delivery safety at home for recovering COVID-19 patients. | Retrospective observational study (UK, 147 patients). | Virtual ward with ambulatory or continuous oxygen. | Demonstrated safety with low readmission and mortality; feasible for selected patients. | Moderate quality; clinically relevant outcomes, but single-centre observational design and selection bias limit generalisability. |
Zychlinski et al., 2024 | To compare outcomes of telemedicine-controlled hospital-at-home vs in-hospital care. | Retrospective matched study (COVID and non-COVID patients). | Telemedicine-supported hospital-at-home (HAH). | Reduced LOS by 2 days; lower 30-day mortality (COVID and non-COVID); no increase in readmissions. | High quality, matched controls, and clear outcome reporting; limited by retrospective design. |
Chang et al., 2021 | To examine the equity implications of rapid telehealth adoption in primary care during COVID-19. | Survey of small practices in NYC (April–June 2020). | Telehealth (video, phone, portals). | Providers in high-vulnerability areas relied on phones rather than video; inequities in access persisted, and policy changes were needed to address them. | Moderate quality; rigorous analysis of equity, but survey bias and lack of clinical outcomes reduce applicability. |
Morís et al., 2024 | To improve clinical decision-making via AI-based multimodal data fusion for COVID-19 risk estimation. | Retrospective study using EHR + imaging (chest X-rays). | Multimodal AI risk prediction model. | High predictive accuracy (AUC ~0.84 hospitalisation; ~0.83 mortality); imaging features useful for hospitalisation; clinical features useful for mortality. | High quality; strong modelling and multimodal dataset, though reliance on imaging limits scalability in low-resource settings. |
Van Goor et al., 2021 | To test the feasibility of remote hospital care for recovering COVID-19 patients. | Randomised controlled trial (62 patients). | Telemedicine-supported hospital care at home (oxygen + monitoring). | Safe and feasible; Reduced index LOS but no improvement in 30-day hospital-free days; increased hospital responsibility days. | Moderate quality: RCT with clear outcomes, but a small sample size and lack of blinding reduce certainty. |
Table 1(B). Contextual sources.
Author/Year | Aim of the Study | Methodology | Treatment / Intervention | Findings of the Study | Quality Appraisal Outcome |
Gentili et al., 2022 | Evaluate the cost-effectiveness of digital health interventions (DHIs). | Systematic review of 35 economic evaluations. | Various DHIs: telehealth, mHealth apps, remote monitoring. | Most DHIs were cost-effective/cost-saving; major heterogeneity in methods and outcomes limited comparability. | High quality; robust RoB assessment and appropriate meta-analysis; minor heterogeneity limitations. |
Tan et al., 2024 | Assess the impacts of RPM on safety, adherence, QoL, and costs. | Systematic review of 29 studies. | Remote patient monitoring during hospital–home transition. | RPM improved safety, adherence, functional outcomes, and reduced readmissions, LOS, and non-hospitalisation costs. | Moderate quality; clear recruitment and outcomes, but no control group; confounder adjustment limited. |
Shi et al., 2024 | Evaluate inpatient-level care at home (virtual wards & HITH). | Systematic review + meta-analysis (69 studies). | Virtual wards / hospital-at-home (low- and high-tech models). | Similar or reduced readmissions; uncertain mortality effects; limited cost-effectiveness data for high-tech models. | Moderate quality; broad search but inconsistent reporting; completeness of included studies unclear. |
Po et al., 2024 | Test the efficacy of post-discharge remote monitoring. | Prospective cohort (41 high-risk patients). | Daily digital monitoring + weekly consultations. | Significant reduction in readmissions and ED visits at 3–6 months; feasible and beneficial for high-risk groups. | High-quality qualitative integration; quantitative arm descriptive with limited confounder control. |
Taylor et al., 2021 | Determine if RPM reduces acute care use. | Systematic review (91 studies). | Remote physiological monitoring across chronic diseases. | RPM reduced hospitalisations and LOS in ~50% of studies; mixed effects on ED use. | High quality for an opinion paper; strong logic and credible expertise; JBI criteria fully met. |
Vo et al., 2025 | Evaluate virtual hospital-in-the-home implementation. | Mixed-methods RE-AIM framework. | COVID-19 virtual HITH service. | Safe, feasible, and saved 16,651 bed-days; barriers included staffing, infrastructure, and the demands of rapid implementation. | High methodological quality; transparent reporting; acknowledges limitations. |
Oliver, 2023 | Critically examine NHS virtual ward policies. | Commentary/policy analysis. | National virtual ward programme (UK). | Warns that the NHS may be overpromising; evidence base is inconsistent; terminology is confusing; and workforce strain risks. | Good quality for scoping design; no RoB assessment (appropriate for scoping). |
Benedetto et al., 2023 | Examine analytical frameworks for digital health economic evaluations. | Methodological systematic review. | Economic evaluation frameworks (CEA, CUA, CCA). | QALY-based models are often insufficient; recommends broader, multi-dimensional frameworks for DHIs. | High MMAT quality; strong triangulation; divergence handling partially reported. |
Islam et al., 2025 | Assess the economic consequences of telehealth in older adults needing unplanned care. | Scoping review (8 studies). | Various telehealth modalities. | Most studies reported cost savings or lower costs; however, the quantity of evidence remains limited and heterogeneous. | Not appraisable; protocol only; lacks empirical appraisal criteria. |
Yahya et al., 2025 | Develop logic & dark logic models for virtual wards. | Document analysis + interviews + focus group. | UK hospital-at-home/virtual ward models. | Identified success factors (clinical buy-in, communication) and risks (inequity, duplication, system strain). | Moderate quality; heterogeneity high; some missing studies likely (Can’t tell). |
Santos et al., 2025 | Map methodologies for DHT economic assessment. | Scoping review protocol. | Frameworks for evaluating digital health technologies. | Outlines gaps in current economic assessment tools; emphasises the need for tailored evaluation models. | High quality; strong meta-analytic methods; minor heterogeneity limitations. |
Rockey-Bartlett et al., 2025 | Examine RPM effects in cancer patients. | Systematic review (9 studies). | RPM for oncology symptom and vital monitoring. | RPM reduced hospitalisations, LOS, and ED visits across several studies; however, evidence remains limited due to heterogeneity. | Low–moderate quality; confounders not controlled; precision limited due to retrospective design. |
Alansari et al., 2025 | Evaluate the impact on paediatric emergencies. | Systematic review + meta-analysis (23 studies). | Telemedicine for paediatric emergency/urgent care. | Reduced mortality and LOS; no significant change in admission rates. | High quality; clear appraisal; heterogeneity reduced comparability. |
Jalilian et al., 2024 | Assess the economic sustainability of a UK virtual ward. | Retrospective longitudinal study. | Hospital virtual ward vs standard inpatient care. | Freed hospital capacity, but the cost per bed-day is higher than inpatient care; frailty/CKD worsened outcomes. | High quality; rigorous RCT inclusion; evidence certainty low–moderate. |
Zakiyah et al., 2024 | Review the cost-effectiveness of DHIs for heart failure. | Systematic review. | Telemonitoring, telehealth, digital self-care tools. | Most interventions dominated standard care (cost-saving + more effective); methodological variation persists. | High quality; large sample size; risk of bias varied, but methods were strong. |
Smedslund et al., 2025 | Analyse RPM effects in noncommunicable diseases. | Systematic review + meta-analysis (40 RCTs). | Digital physiological monitoring. | RPM modestly reduced hospitalisations and LOS; no effect on ED visits; low–moderate certainty evidence. | High quality; robust RoB assessment and appropriate meta-analysis; minor heterogeneity limitations. |
Jansen et al., 2025 | Evaluate the impact of device-based monitoring on hospital service use. | Systematic review of RCTs (116 trials). | Device-based monitoring (implanted, wearable, mobile). | 72% of trials showed reduced hospital service use; the strongest effects were with patient-driven or 24/7-supported models. | Moderate quality; clear recruitment and outcomes, but no control group; confounder adjustment limited. |
Remote patient monitoring (RPM) has equally heterogeneous results. Meta-analyses indicate slight but significant decreases in hospitalisation and length of stay in the case of chronic and high-risk populations, especially where adherence and clinical monitoring are good [21,22]. Future evidence by Po et al. [22] supports the idea that post-discharge RPM can have the most significant impact on high-risk populations with a well-organised escalation protocol, with wider systematic reviews also indicating a mixed impact on emergency department use [20]. In specialised populations studied by Rockey-Bartlett et al. [26], including oncology, the potential is demonstrated, but lacks large sample sizes, measures are not homogenous, and confounding is not fully adjusted. Greater adherence has been seen in device-based models of monitoring: Jansen et al. [24] found that 72 per cent of RCTs reported less hospital use with patient-activated or continuously supported monitoring, which suggested that engagement and responsiveness are factors of success. A combination of these studies highlights the idea that the outcomes of RPM are not determined by the used technology, but rather by patient selection, the intensity of supervision, and the quality of engagement.
Together, these results suggest that VWs do not necessarily help to save money, and they cannot be universal but must be applied in specific ways to digital infrastructure and sustainable workforce models.
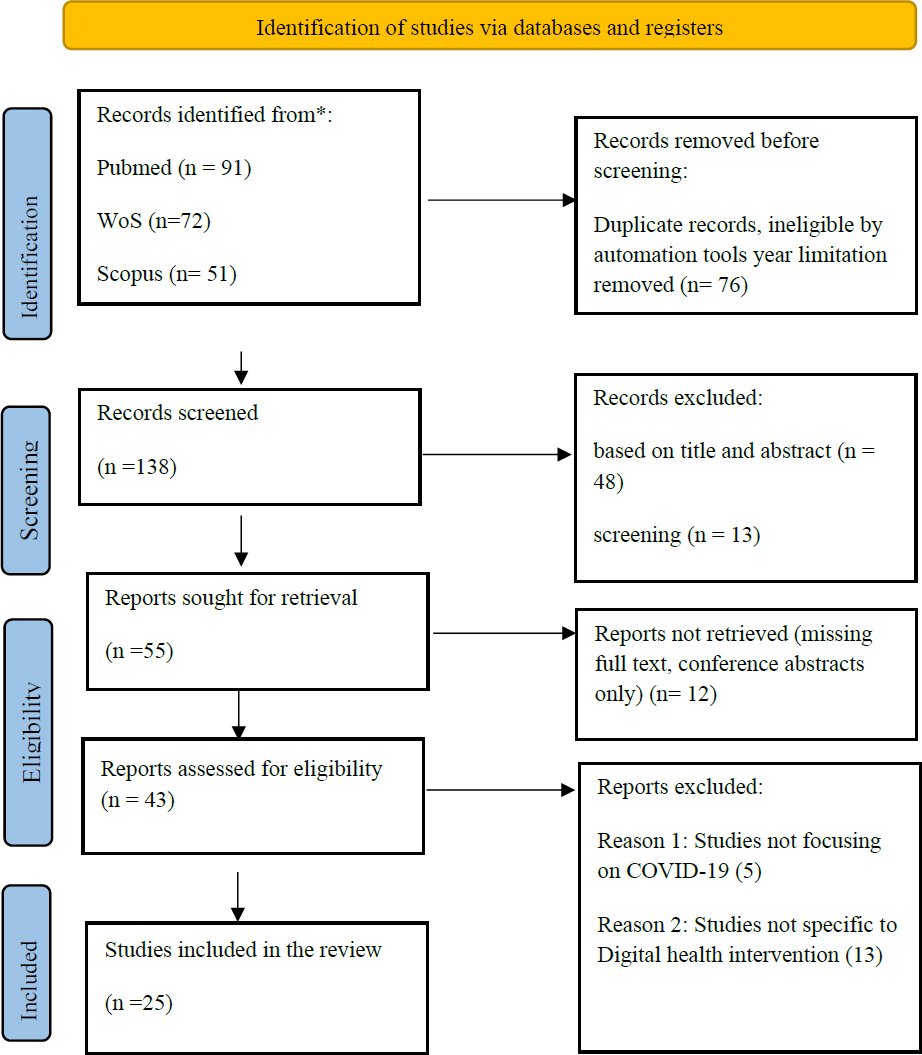
Fig. (1). PRISMA Flow Diagram.
3.3. Telehealth and Equity in Access to Care
During the COVID -19 pandemic, the blistering development of telehealth services revealed glaring disparities in access and the use of modalities. Chang et al. [4] showed that the rate of telephone encounters was higher in high-vulnerability communities (41.7 %), compared to video consultations, which were more suitable in areas with wealth (33.7 % versus 18.7 %), due to the presence of such barriers as access to devices, computer literacy, and broadband access. Conversely, Sankaranarayan et al. [16] found that heart failure virtual wards (HFVW), where telehealth was integrated, significantly lowered rehospitalisations and death rates at various follow-up stages. According to the authors, the clinical risk consistently was lower than those of standard care, the clinical risk odds ratio of 1-month readmission was 0.30 (95 CI 0.20 -0.50) and odds ratio mortality 0.26 (95 CI 0.14-0.48), that is, equity-neutral benefits have a higher possibility of success in the environments that are furnished with the infrastructure of specialists and intensive monitored environments. Ward et al. [8] reported low mortality and readmission rates in an oxygen therapy virtual ward in the UK, with limitations put on the study by its single-centre nature. On the contrary, Beaney et al. [13] found that the Oximetry @home programme in England did not provide any mortality benefit and had reduced overall enrolment (2.5 per cent), thus negatively impacting the outcome. In addition, van Goor et al. [14] discovered that hospital care using telemedicine reduced inpatient length of stay and did not result in more hospital-free days, casting doubt on the ability of telehealth to alleviate the impact of acute-care burdens. Yahya et al. [31] opined that digital interventions have the potential to contribute to inequalities, redundancy in services, and overtaxing community providers without lowering hospital demand. Such apprehensions resonate with those depicted by Chang et al. [4], who found that more reimbursable telehealth modalities were lower among high-income groups, which then consolidated access inequities. Together with the risk of overstating performance in policy discourse, these findings also point to the fact that digital health interventions cannot be treated as a neutral solution to problems; they can further increase inequity unless accompanied by digital literacy, inclusive enrolment policies, and infrastructure provision.
3.4 Economic Sustainability & Evaluation Frameworks for DHIs
The interpretation of economic findings was done narratively based on a predefined health-system approach that targeted direct service delivery costs. Since no formal economic extraction grid or meta-analytic cost synthesis was performed, these results ought to be viewed as organised narrative economical observations as opposed to an economic synthesis. Even though secondary systematic reviews have shown that numerous digital health interventions emerge to be cost-effective or cost-saving [27, 28], substantial methodological inconsistencies and limited reporting of implementation and operating costs restrict the transferability of findings across healthcare systems. In this review, economic evidence was interpreted using a predefined and consistent evaluation framework. The primary analytical perspective adopted was that of the health-care system, focusing on resource use and expenditure borne by providers rather than wider societal costs. A short-to-medium time horizon was applied, corresponding to the follow-up periods most frequently reported by the included studies, typically 30-day and 6–12-month post-implementation windows.
Cost components were operationalised as direct service delivery and implementation inputs, including staff time for monitoring and virtual consultations, digital devices and software infrastructure, training and technical support, hospital admissions avoided, emergency department attendances, and bed-days saved. In line with methodological critiques of relying solely on QALYs [33], outcome valuation in this review did not privilege health-utility measures alone. Instead, economic outcomes were extracted and synthesised primarily as cost per admission avoided, cost per readmission prevented, and cost per bed-day saved, reflecting resource allocation and operational efficiency more directly.
The need for multidimensional valuation of digital interventions, including workforce effects, continuity of care, and equity considerations, is further emphasised by scoping reviews and protocols [29,34,35]. Consequently, policy-relevant interpretations in this review are grounded in structured cost components and clearly defined time horizons, rather than in simplified or naïve cost-saving assumptions, thereby supporting context-specific economic decision-making in post-pandemic healthcare systems.
4. Discussion
The resulting discussion is an interpretation of findings based on the input process outcome and output framework to reduce the overlap of themes. The contrasts in heterogeneity among studies were organised patternically and not by chance. The effects of the intervention were different based on the intensity of implementation, structure, and health-system context. Low-technology methods, like telephone follow-up and pulse-oximetry monitoring, were more scalable but yielded mixed results in the presence of ineffective enrolment and escalation pathways. Greater-intensity models, such as specialist-devoted virtual wards and hospital-at-home programmes, fared better in well-resourced systems. In general, digital care models seemed the most successful when three conditions were combined: a deep level of patient engagement, formalised escalation routes, sufficient staffing and digital infrastructure, and generalisability to lower-resource contexts was unclear.
The discussion of the results highlights a massive difference between the effectiveness of remote monitoring patterns and virtual wards. Even though Beaney et al. [13] and Ward et al. [8] proved the possibility of taking a load off hospitals by introducing national programmes and single-centre trials, they also show serious limitations. According to Beaney et al. [13], there was no statistically significant mortality reduction and an increased admission rate in a population-level assessment, which was mainly due to low enrolment and unbalanced programme delivery by region. The low involvement and inefficient enrolment in national rollouts hindered the ability of the programme to achieve quantifiable benefits, considering the dilution effect of a 2.5 enrolment rate. Policies that are equity-conscious and that broaden telehealth service coverage are thus necessary to realise digital health potential to enhance care delivery. These factors remind the reader of the significance of implementation fidelity, a key success factor of digital health programmes. Ward et al. [8], on the contrary, had a more specific virtual ward model, which gained promising outcomes such as low mortality and readmission rates. These favourable outcomes were almost inevitably due to the targeted, clinically selected patient cohort, high involvement, and standardised intervention protocols. This comparison thus highlights the need for a patient selection and engagement strategy to achieve the required results. In addition, findings were reported in highly structured models by van Goor et al. [14] and Zychlinski et al. [15].
4.1. Inputs
The results of the study point to one key issue: even though the telehealth modalities hold significant potential to enhance patient outcomes and benefits, they also reveal deep-rooted inequities that determine patient access to care. Chang et al. [4] found significant differences in telehealth adoption, with low-income and vulnerable groups overrepresented among those who rely on telephone consultations, which are less effective than video-based consultations. This digital divide contributes to the existing disparities in healthcare, particularly among marginalised groups, which typically lack the necessary infrastructure and resources to receive higher-quality telemedicine. Ward et al. [8] and Sankaranar et al. [16] showed that well-planned, specialist-led interventions can improve patient care without overwhelming hospitals, thereby motivating serious clinical outcomes associated with telehealth interventions. However, these programmes were applied in resource-heavy environments, which makes their applicability to low- and middle-income countries (LMICs) questionable as the digital infrastructure is not currently widely available. The success of hospital-at-home models had also been reported by Zychlinski et al. [15]. However, they emphasised that these results were possible only in well-resourced settings, thus challenging their applicability in low-resource healthcare systems. The efficacy of the digital health interventions is also critical for enrolment and standardisation, as Beaney et al. [13] further emphasised. The Oximetry @home programme failed to achieve its desired results due to the lack of engagement of the participants. This is a severe constraint in large-scale implementations; engagement policies should be robust enough to implement desired interventions and achieve their potential. Moreover, van Goor et al. [14] established that inpatient length of stay can indeed be reduced by remote hospital care. However, these interventions do not necessarily lead to a net decrease in overall healthcare demand, which makes the effectiveness aspect of telehealth complicated. All these results point to the fact that digital health implementation is facing a set of structural challenges that include digital literacy, broadband connectivity, and device affordability, among others. There is therefore need to have equity-based policies which increase the range of telehealth services to realise the potential of digital health of enhancing care delivery.
It has been shown that predictive modelling and artificial intelligence in healthcare can be useful in terms of allocating resources. However, its shortcomings shed some light on the intricacy of the real-world implementation. Tariq et al. [17] have demonstrated that predictive accuracy is better with full datasets, i.e., medical histories, demographics, and other variables of interest of the patients. However, the application of the model in a single healthcare system in the U.S. cities prompts the issue of the model applicability to low-resource or rural locations where the number of patients differs, and the ability to retrieve all clinical records is restricted. Besides, the study notes the value of big data that are heterogeneous to enhance generalisability, which is often impossible with limited digital infrastructure and ill-conceived data-collection systems in most healthcare systems, particularly in low- and middle-income countries (LMICs). Moris et al. [18] used a technologically superior model for multimodal information fusion, including clinical history and imaging data, which improved predictive performance. However, high-tech imaging capabilities are highly demanding to rely on, particularly in resource-deprived contexts where imaging facilities are scarce. The inherently opaque so-called black-box character of deep-learning models also brings about the question of transparency; clinicians will be challenged in understanding the logic underlying the algorithmic predictions, thus undermining trust and hampering the smooth incorporation of such technologies into clinical decision-making procedures. All of these studies combined highlight the importance of using AI, which can potentially improve resource utilisation and patient outcomes; however, special attention should be paid to the issues of data accessibility, model transparency, and generalisability. Policy makers should ensure fair availability of the necessary data to train AI-based models and ensure that they develop interpretable AI models that can reduce bias and build trust.
4.2. Processes
One of the current trends in digital health interventions is that implementation fidelity mediates the efficacy. In England, the national COVID-19 Oximetry at home programme, for example, did not reduce mortality because of poor enrolment rates and inconsistent implementation practices [7]. On the other hand, positive results were recorded in a single-centre virtual ward where structured engagement protocols were used by Ward et al. [8]. However, even the most strictly developed models cannot scale without a strong systemic infrastructure that makes uptake possible. In comparison, the models with specialist leadership, like the Heart Failure Virtual Ward, achieved higher results Sankaranarayan et al. [16] however, only in the resource-abundant settings. Van Goor et al. [14] and Zychlinski et al. [15] evidence supported the view that results are dependent on the contextual preparedness rather than on the type of intervention. The artificial-intelligence applications [17] [18] demonstrated the predictive accuracy but were not without concern about bias and generalisability.
4.3. Outputs
The digital models of care, such as virtual wards, hospital at home, and remote patient monitoring, have shown potential but highly contextual effects on hospital utilisation. Though in large comparative analyses, shorter length of stay and no or decreased readmission rates are observed in well-resourced environments [19, 20, 22, 23,24, 25,30]. These models have not always been shown to be effective, and their implementation has been proposed to redistribute as well as to lower aggregate demand in case of insufficient staffing or escalation routes [14,25]. Similarly, post-discharge RPM has the highest DEN, high-risk, actively monitored cohorts. Po et al. [22] and Tan et al. [23] have the highest certainty, although heterogeneity, small-sample studies, and weak control of confounders limit such certainty in specialised populations such as oncology [26]. Stable increases are achieved with the implementation of continuous or patient-triggered device monitoring, a consistent decrease in hospital utilisation observed across RCTs by Jansen et al. [24], which is important to stress that the level of engagement, but not the adoption of technology per se, is what leads to improvements. Economic results also point to the fact that cost-effectiveness cannot be presumed, since capacity gains might go along with increased operational costs when there are suboptimal occupancy, staffing, and digital infrastructure, and the methodological inconsistency prevents generalisability [27,28, 32]. New models say that there is a need to have multidimensional, equity-sensitive metrics other than QALY, which include workforce impact, digital access, and system duplication [29, 33,34, 35].
4.4. Outcomes and System Resilience
The recent national and international guidelines support the fact that virtual wards should be introduced with caution and well-defined patient selection criteria. Policy briefs by the UK point to the fact that the experiences of patients and carers are still positive, but the expectations are usually higher than what clinical potentials can achieve [36,37,38]. Frail elderly assessment demonstrates that it may be beneficial, but with great differences in safety and staff preparedness [39, 40, 41,42,43]. Greater studies warn that there must not be assumptions of universal cost-saving that lack standardised economic data [33, 40]. The results of large-scale RPM studies also indicate fewer admissions and caution that the results are reliant on the resources and digital literacy, as well as monitoring capacity [37,41].
Digital health projects must be transformed to go beyond the response-based, ad hoc, and crisis-driven initiatives to systematic inclusion in existing care pathways. Remote-monitoring, virtual-ward, and artificial-intelligence devices are to be implemented as supplements to traditional methods of care, and they need clear instructions concerning the route of escalation, inclusion criteria, and the individuals who will work with the patients, as well as multidisciplinary team coordination. Future digital health implementations are to be patient-centred and standardised. Policymakers must set minimum requirements on the quality of digital tracking, the equivalence of telehealth reimbursement, and ongoing clinician training to ensure successful digital monitoring. Transparency should maximise trust in AI-driven decision support. New models must prefigure an explainable AI (XAI) system, constantly testing in various groups, and strictly controlled. The ethical regulation of privacy, consent, and accountability must be enforced to avoid harm and reduce the disparities caused by deployment. Lastly, digital-health interventions are to be included in the strategies to improve system resiliency. Research [15] [16] has shown that hospital-at-home and telehealth programmes can potentially reduce acute care demand in times of crisis. The tools cannot be applied only during the pandemic but should be used to be ready to the shocks in the future like ageing, a load of chronic diseases, and health crisis in a climate. The post-pandemic world teaches that despite its potentially powerful nature, digital-health intervention is not transformative: its effectiveness lies on equity, fidelity, context, and ethical integration. It is believed that future healthcare will be hybrid systems where digital and traditional elements are bound together to guarantee efficiency, capacity, equity, transparency, and resilience. Because the included studies employed heterogeneous designs, outcome definitions, and analytical approaches, direct quantitative comparison of effect sizes was not appropriate and may limit the interpretability of cross-study differences. Cumulatively, this review suggests that digital care models can produce a capacity substitution in conditions of implementation fidelity and workforce elasticity.
4.5. Future directions: AI-enabled planning and decision support
AI-based models showed promising predictive performance for hospitalisation and mortality, particularly when multimodal datasets were available, but their role in the current evidence base remains primarily anticipatory rather than implementation-proven. Their contribution is therefore best interpreted as a future direction for digital system planning rather than as established evidence of service-level resource substitution. Key concerns remain data bias, generalisability, infrastructure dependence, and model transparency.
Limitations
The limitations of this review are a few. First, the evidence included was heterogeneous in design, intensity of interventions, and reporting outcomes, restricting a direct comparison between studies and precluding meta-analysis. Second, publication bias and a restriction to the English language might also have limited the evidence base. Third, the protocol of review was not prospectively registered. Fourth, despite primary studies informing the overall synthesis, a few secondary and contextual sources were selected to map information, which arguably minimises interpretive purity but may improve policy relevance. Lastly, since most studies have been conducted in well-resourced systems, we should be careful to generalise conclusions to less-resourced settings or to the future after the pandemic.
Conclusion
The review indicates that virtual wards, hospital-at-home care, and remote services could reduce length of stay and eliminate avoidable readmissions, especially when they focus on high-risk populations and have well-defined escalation routes. Nevertheless, not all of them are effective, and the inadequately-resourced implementation would not decrease the healthcare demand but could increase the digital inequalities instead. The economic performance might still be bright and unstable because of fluctuating costing systems and immeasurable personnel and infrastructure strains. Thus, cost-saving solutions in the form of digital care models should not be scaled automatically but should be applied selectively with a strong evaluation, equity protection, and standardised clinical and economic outcomes. Most consistent clinical and resource-related benefits were observed in studies rated as high or moderate quality. Evidence derived from low-quality studies was not used to inform policy or implementation recommendations. Lastly, this review suggests that digital care models do not automatically reduce system pressure; rather, they generate meaningful capacity substitution only when implementation fidelity, workforce elasticity, and supportive digital infrastructure are present.
Funding
None.
Declaration of Competing Interests
The authors declare no competing interests. All authors contributed to the manuscript’s conception, design, and writing.
References
[1] Jat AS, Grønli TM. Harnessing the digital revolution: a comprehensive review of mHealth applications for remote monitoring in transforming healthcare delivery. In International Conference on Mobile Web and Intelligent Information Systems 2023 Aug 3 (pp. 55-67). Cham: Springer Nature Switzerland. https://arxiv.org/pdf/2408.14190
[2] Getachew E, Adebeta T, Muzazu SG, Charlie L, Said B, Tesfahunei HA, Wanjiru CL, Acam J, Kajogoo VD, Solomon S, Atim MG. Digital health in the era of COVID-19: Reshaping the next generation of healthcare. Frontiers in public health. 2023 Feb 15; 11:942703. https://doi.org/10.3389/fpubh.2023.942703
[3] Periáñez Á, Fernández Del Río A, Nazarov I, Jané E, Hassan M, Rastogi A, Tang D. The digital transformation in health: How AI can improve the performance of healthcare systems. Healthcare systems & Reform. 2024 Dec 17;10(2):2387138. https://doi.org/10.1080/23288604.2024.2387138
[4] Chang JE, Lai AY, Gupta A, Nguyen AM, Berry CA, Shelley DR. Rapid transition to telehealth and the digital divide: implications for primary care access and equity in a post‐COVID era. The Milbank Quarterly. 2021 Jun;99(2):340-68. https://onlinelibrary.wiley.com/doi/pdfdirect/10.1111/1468-0009.12509?casa_token=rml2GzKuiYMAAAAA:JfpQU8Y2MJ9OLVE72aeoGDjZTMFDPn1ze7LuWRTr5uQDFyHqijVu301jFPH0AOqw4nGY1WFUy8ivc1U
[5] Ezeamii VC, Okobi OE, Wambai-Sani H, Perera GS, Zaynieva S, Okonkwo CC, Ohaiba MM, William-Enemali PC, Obodo OR, Obiefuna NG. Revolutionising healthcare: how telemedicine improves patient outcomes and expands care access. Cureus. 2024 Jul 5;16(7). https://doi.org/10.7759/cureus.63881
[6] Härkönen H, Lakoma S, Verho A, Torkki P, Leskelä RL, Pennanen P, Laukka E, Jansson M. Impact of digital services on healthcare and social welfare: An umbrella review. International journal of nursing studies. 2024 Apr 1; 152:104692. https://doi.org/10.1016/j.ijnurstu.2024.104692
[7] Li J, Me RC, Ahmad FA, Zhu Q. Investigating the application of IoT mobile app and healthcare services for diabetic elderly: A systematic review. PLoS One. 2025 Apr 15;20(4):e0321090. https://doi.org/10.1371/journal.pone.0321090
[8] Ward, T.J., Mavilakandy, A., Danns, J., Tsaknis, G. and Reddy, R.V., 2022. Outcomes from a virtual ward delivering oxygen at home for patients recovering from COVID-19: a real-world observational study. Clinical Medicine, 22(3), pp.197-202. https://doi.org/10.7861/clinmed.2021-0512
[9] Zrubka Z, Champion A, Holtorf AP, Di Bidino R, Earla JR, Boltyenkov AT, Tabata-Kelly M, Asche C, Burrell A. The PICOTS-ComTeC framework for defining digital health interventions: an ISPOR special interest group report. Value in Health. 2024 Apr 1;27(4):383-96. https://doi.org/10.1016/j.jval.2024.01.009
[10] Siddaway AP, Wood AM, Hedges LV. How to do a systematic review: a best practice guide for conducting and reporting narrative reviews, meta-analyses, and meta-syntheses. Annual review of psychology. 2019 Jan 4;70(1):747-70. https://doi.org/10.1146/annurev-psych-010418-102803
[11] Parums DV. Review articles, systematic reviews, meta-analyses, and the updated Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines. Medical science monitor: international medical journal of experimental and clinical research. 2021 Aug 23;27:e934475-1. https://doi.org/10.12659/MSM.934475
[12] McKenna-Plumley PE, Groarke JM, Turner RN, Yang K. Experiences of loneliness: a study protocol for a systematic review and thematic synthesis of qualitative literature. Systematic Reviews. 2020 Dec 6;9(1):284. https://doi.org/10.1186/s13643-020-01544-x
[13] Beaney, T., Clarke, J., Alboksmaty, A., Flott, K., Fowler, A., Benger, J., Aylin, P.P., Elkin, S., Neves, A.L., and Darzi, A., 2022. Population-level impact of a pulse oximetry remote monitoring programme on mortality and healthcare utilisation in people with COVID-19 in England: a national analysis using a stepped wedge design. Emergency Medicine Journal, 39(8), pp.575-582. https://doi.org/10.1136/emermed-2022-212378
[14] van Goor HM, Breteler MJ, van Loon K, de Hond TA, Reitsma JB, Zwart DL, Kalkman CJ, Kaasjager KA. Remote hospital care for recovering COVID-19 patients using telemedicine: a randomised controlled trial. Journal of Clinical Medicine. 2021 Dec 17;10(24):5940. https://doi.org/10.3390/jcm10245940
[15] Zychlinski N, Fluss R, Goldberg Y, Zubli D, Barkai G, Zimlichman E, Segal G. Tele-medicine-controlled hospital at home is associated with better outcomes than hospital stay. Plos one. 2024 Aug 19;19(8):e0309077. https://doi.org/10.1371/journal.pone.0309077
[16] Sankaranarayanan R, Rasoul D, Murphy N, Kelly A, Nyjo S, Jackson C, O’Connor J, Almond P, Jose N, West J, Kaur R. Telehealth‐aided outpatient management of acute heart failure in a specialist virtual ward compared with standard care. ESC Heart Failure. 2024 Dec;11(6):4172-84. https://doi.org/10.1002/ehf2.15003
[17] Tariq A, Celi LA, Newsome JM, Purkayastha S, Bhatia NK, Trivedi H, Gichoya JW, Banerjee I. Patient-specific COVID-19 resource utilisation prediction using fusion AI model. NPJ digital medicine. 2021 Jun 3;4(1):94. https://doi.org/10.1038/s41746-021-00461-0
[18] Morís DI, de Moura J, Marcos PJ, Rey EM, Novo J, Ortega M. Efficient clinical decision-making process via AI-based multimodal data fusion: A COVID-19 case study. Heliyon. 2024 Oct 30;10(20). https://doi.org/10.1016/j.heliyon.2024.e38642
[19] Shi C, Dumville J, Rubinstein F, Norman G, Ullah A, Bashir S, Bower P, Vardy ER. Inpatient-level care at home delivered by virtual wards and hospital at home: a systematic review and meta-analysis of complex interventions and their components. BMC Medicine. 2024 Apr 2;22(1):145. https://doi.org/10.1186/s12916-024-03312-3
[20] Taylor ML, Thomas EE, Snoswell CL, Smith AC, Caffery LJ. Does remote patient monitoring reduce acute care use? A systematic review. BMJ Open. 2021 Mar 1;11(3):e040232. http://dx.doi.org/10.1136/bmjopen-2020- 040232
[21] Smedslund G, Østerås N, Hestevik CH. Effects of remote patient monitoring on health care utilisation in patients with noncommunicable diseases: Systematic review and meta-analysis. JMIR mHealth and uHealth. 2025 Oct 1;13:e68464. https://doi.org/10.2196/68464
[22] Po HW, Chu YC, Tsai HC, Lin CL, Chen CY, Ma MH. Efficacy of remote health monitoring in reducing hospital readmissions among high-risk postdischarge patients: prospective cohort study. JMIR Formative Research. 2024 Sep 13;8:e53455. https://doi.org/10.2196/53455
[23] Tan SY, Sumner J, Wang Y, Wenjun Yip A. A systematic review of the impacts of remote patient monitoring (RPM) interventions on safety, adherence, quality-of-life, and cost-related outcomes. NPJ Digital Medicine. 2024 Jul 18;7(1):192. https://doi.org/10.1038/s41746-024-01182-w
[24] Jansen AJ, Peters GM, Kooij L, Doggen CJ, van Harten WH. Device-based monitoring in digital care and its impact on hospital service use. npj Digital Medicine. 2025 Jan 8;8(1):16. https://doi.org/10.1038/s41746-024-01427-8
[25] Oliver D. David Oliver: With virtual wards, NHS England is overpromising once again. bmj. 2023 Nov 29;383. https://doi.org/10.1136/bmj.p2787
[26] Rockey-Bartlett C, Morelli J, Coffel M, Geracitano J, Lafata JE, Khairat S. Effect of remote patient monitoring on healthcare use among patients with cancer: A systematic review. Digital Health. 2025 Oct;11:20552076251384220.
[27] Gentili A, Failla G, Melnyk A, Puleo V, Tanna GL, Ricciardi W, Cascini F. The cost-effectiveness of digital health interventions: a systematic review of the literature. Frontiers in public health. 2022 Aug 11;10:787135.
[28] Zakiyah N, Marulin D, Alfaqeeh M, Puspitasari IM, Lestari K, Lim KK, Fox-Rushby J. Economic evaluations of digital health interventions for patients with heart failure: systematic review. Journal of medical Internet research. 2024 Apr 30;26:e53500. https://doi.org/10.2196/53500
[29] Islam F, Sowada C, Siqeca F, Sabbe M, Milisen K, Kocot E. Economic evaluations and financial consequences related to the use of telehealth services in older adults seeking unplanned care: a scoping review. BMC Digital Health. 2025 Sep 17;3(1):59. https://doi.org/10.1186/s44247-025-00199-9
[30] Vo LK, Carter HE, McPhail SM, McGowan K, Wallis S, Atkinson K, Allen MJ. Implementation of a Virtual Hospital in the Home Service for Patients With COVID-19 in Queensland, Australia: Mixed Methods Evaluation Using the RE-AIM Framework. Journal of Medical Internet Research. 2025 Sep 19;27:e73749. https://doi.org/10.2196/73749
[31] Yahya F, Cooper M, Nazar H. Hospital at home (virtual wards): developing a logic model and dark logic model. BMC Health Services Research. 2025 May 17;25(1):714. https://doi.org/10.1186/s12913-025-12872-w
[32] Jalilian A, Sedda L, Unsworth A, Farrier M. Length of stay and economic sustainability of virtual ward care in a medium-sized hospital of the UK: a retrospective longitudinal study. BMJ Open. 2024 Jan 1;14(1):e081378. https://doi.org/10.1186/s12913-025-12872-w
[33] Benedetto V, Filipe L, Harris C, Spencer J, Hickson C, Clegg A. Analytical frameworks and outcome measures in economic evaluations of digital health interventions: a methodological systematic review. Medical Decision Making. 2023 Jan;43(1):125-38. https://doi.dox.org/10.1177/0272989X221132741
[34] Santos AR, Sampaio F, Londral AR, Perelman J. Mapping methodologies for economic assessment of digital health technologies: a scoping review protocol. BMJ Open. 2025 Aug 1;15(7):e099933. https://doi.org/10.1136/bmjopen-2025-099933
[35] Alansari AN, Zaazouee MS, Messaoud M, Mani S, Elshanbary AA, Mohamed HY. Impact of telemedicine on mortality, hospital admissions, and length of stay in pediatric emergencies: a systematic review and meta-analysis. International Journal of Emergency Medicine. 2025 Aug 27;18(1):160. https://doi.org/10.1186/s12245-025-00968-3
[36] National Health Service (NHS) — Strategy Unit. Virtual Wards Evaluation: Patients’ and Unpaid Carers’ perspectives. 2025. Available from: https://strategyunitwm.nhs.uk
[37] NHS Confederation. Realising the potential of virtual wards. Policy brief. 2023. Available from: https://www.nhsconfed.org
[38] National Institutes of Health (NIH). Virtual Medicine Wards and Hospital-at-Home Programs. Research & Analysis. NCBI Bookshelf. Available from: https://www.ncbi.nlm.nih.gov/books
[39] Cureus. The effectiveness of virtual wards compared to inpatient beds in frail older patients: systematic review. 2025. Available from: https://www.cureus.com
[40] NHS England; Strategy Unit. Evidence briefs and guidance on virtual wards/hospital-at-home. 2024–2025. Available from: https://ewin.nhs.uk
[41] Liebert; Telemedicine Journal. Impact of a large-scale RPM program on admissions: observational analyses. 2024. Available from: https://www.liebertpub.com
[42] Benedetto V, et al. Digital health interventions economic evaluation: systematic review. 2022. AAM. Available from: University of Central Lancashire repository. https://arxiv.org/pdf/2307.08393
[43] British Geriatrics Society (BGS). Virtual wards and hospital at home for older people: guidance. 2022. Available from: https://www.bgs.org.uk
Licensed
© 2026 Copyright by the Authors.
Licensed as an open access article using a CC BY 4.0 license.
Article Contents Author Ishaq Kalanther1, * 1Jubail Industrial College, Jubail Industrial City, Jubail, Kingdom of Saudi Arabia Article History: Received:
Article Contents Author Mirza Amin ul Haq1 Ngan Thi Luong2 Arsalan Mujahid Ghouri2 1Faculty of Engineering,
Article Contents Author Usama Haider1, * 1Department of Physics, Forman Christian College University, Lahore, Pakistan Article History: Received: 22 September,
Article Contents Author Yazeed Alsuhaibany1, * 1College of Business-Al Khobar, Al Yamamah University, Saudi Arabia Article History: Received: 03 September,
Article Contents Author Gilbert Talaue1, * 1Jubail Industrial College, Al Jubail, Saudi Arabia Article History: Received: 11 November, 2025 Accepted:
Article Contents Author Burhanettin Uysal1, * Maria Malik2 1Department of Healthcare Management, Bilecik Şeyh Edebali University, Turkey Article History: Received:



















 PDF
PDF